In other words, \(\psi^2\) describes the shape and size of an electron's orbital (the shapes you already know). The probability of finding the electron somewhere in space is the square of the wavefunction (\(\psi^2\). The \(\psi\) describes the wave properties of an electron. However, there are an infinite number of values for \(n\) from \(n=1\rightarrow\infty\), so there are also infinite solutions to the Schr ö dinger equation. In other words the allowed solutions are quantized. Some important considerations and limitationsĪlthough it might seem like there could be any value of x, y, and z for the Hamiltonian, these values are limited by the allowed positions of electrons according to \(\psi\), which is limited by integer values of \(n\). The imaginary property we call "spin", up or down Orientation of angular momentum in space, orbital *letters have historical meaning, sharp, principle, diffuse, fundamental This is the angular dependence of the orbital, shape of the orbital For example \(p_x\) lies along the \(x\) axis, while \(p_y\) points in a different direction in space.įor review, a list of the quantum numbers, their values, and meanings are in the table below. The angular variation, \(Y\), depends on the angle with respect to the x, y, and z coordinates, and depends on the quantum numbers \(l\) (the orbital type) and \(m_l\) (the angular momentum, or the specific orbital). Since \(n\) must be an integer, there are only certain allowed values for the solution to the wavefunction.
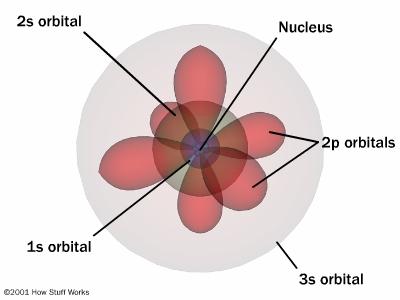
The quantum numbers \(n\) (energy level) and \(l\) (orbital type) define \(R\). These quantum numbers will be discussed more in a later section (2.2.2) The radial variation, \(R\), depends on the electron's distance from the nucleus. The (\(\psi\)) is defined by three of the quantum numbers: \(n\), \(l\), and \(m_l\). If the electron is very far from the nucleus (\(r = \infty\)) then the attractive force, and the potential energy, is zero. When an electron is farther from the nucleus, the potential energy is still negative but with a smaller magnitude, corresponding to a weaker attractive force.

Thus, when an electron is close to the nucleus, the potential energy is a large negative number corresponding to a strong attractive force. And, just as in Coulomb's law, when two opposite charges are attracted to one another, the potential energy of the force is negative. You might notice that this attraction is essentially the same as the electrostatic force defined by Coulomb's law. The potential energy depends on the attractive electrostatic force between the electron and the nucleus.
